 *Complex multivalent vaccines can have production lead times of more than 36 months.ġ. These strong quality requirements involve ad hoc pharmaceutical quality systems, quality assurance measures and procedures, several quality controls at each stage and an adequate infrastructure and separation of activities to guarantee vaccine identity, purity, sterility, efficacy and safety. Vaccine production involves a spectrum of designs that can be based on anything between attenuated organisms and complex compounds produced by molecular biology. Although it can take a long time to produce vaccines, they are not difficult to produce. Moderna is increasing its Covid-19 vaccine production this year, upping the minimum number of doses it expects to make by 20 to 600 million, the company announced Monday. Congress passed An act to regulate the sale of viruses, serums, toxins. Vaccines are made from dead (inactivated) or modified (attenuated live) whole microbes, or from inactivated or recombinant parts of microbes that are responsible for disease (such as toxins or surface proteins). 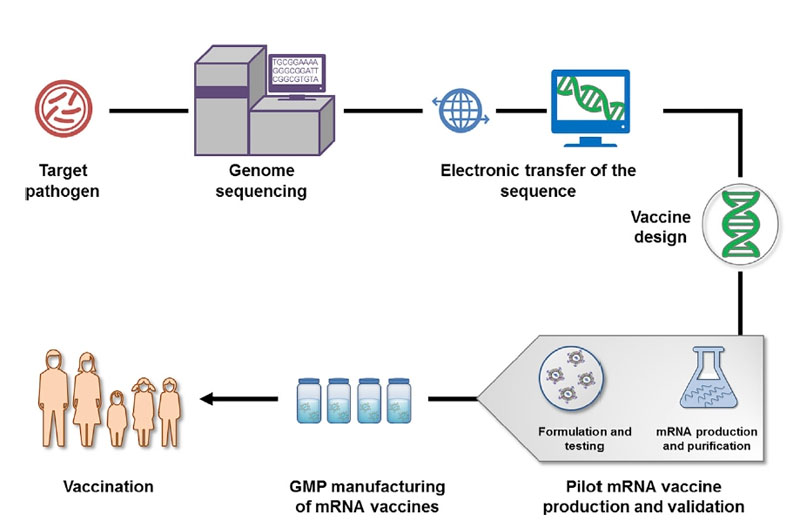 
Successful manufacturing of high-quality vaccines requires international standardization of starting materials, production and quality control testing, and the setting of high expectations for regulatory oversight of the entire manufacturing process from start to finish, all while recognizing that this field is in constant change 1.Īll the components, manufacturing processes, testing methods, their reagents and standards have to comply with the standards defined for Good Manufacturing Practices (GMP). However, no regulation of vaccine production existed. They are then fed to the main manufacturing centres. The quality controls represent up to 70% of the full manufacturing duration. 15 companies in the Netherlands play a key role in the production and global distribution of vaccines for human use. The manufacturing of raw materials and drug substances happens at two centres in the US: in Saint Louis in Missouri and Andover in Massachusetts. Vaccine production in plants is already a fact due to advances of molecular farming (Chapter 4). 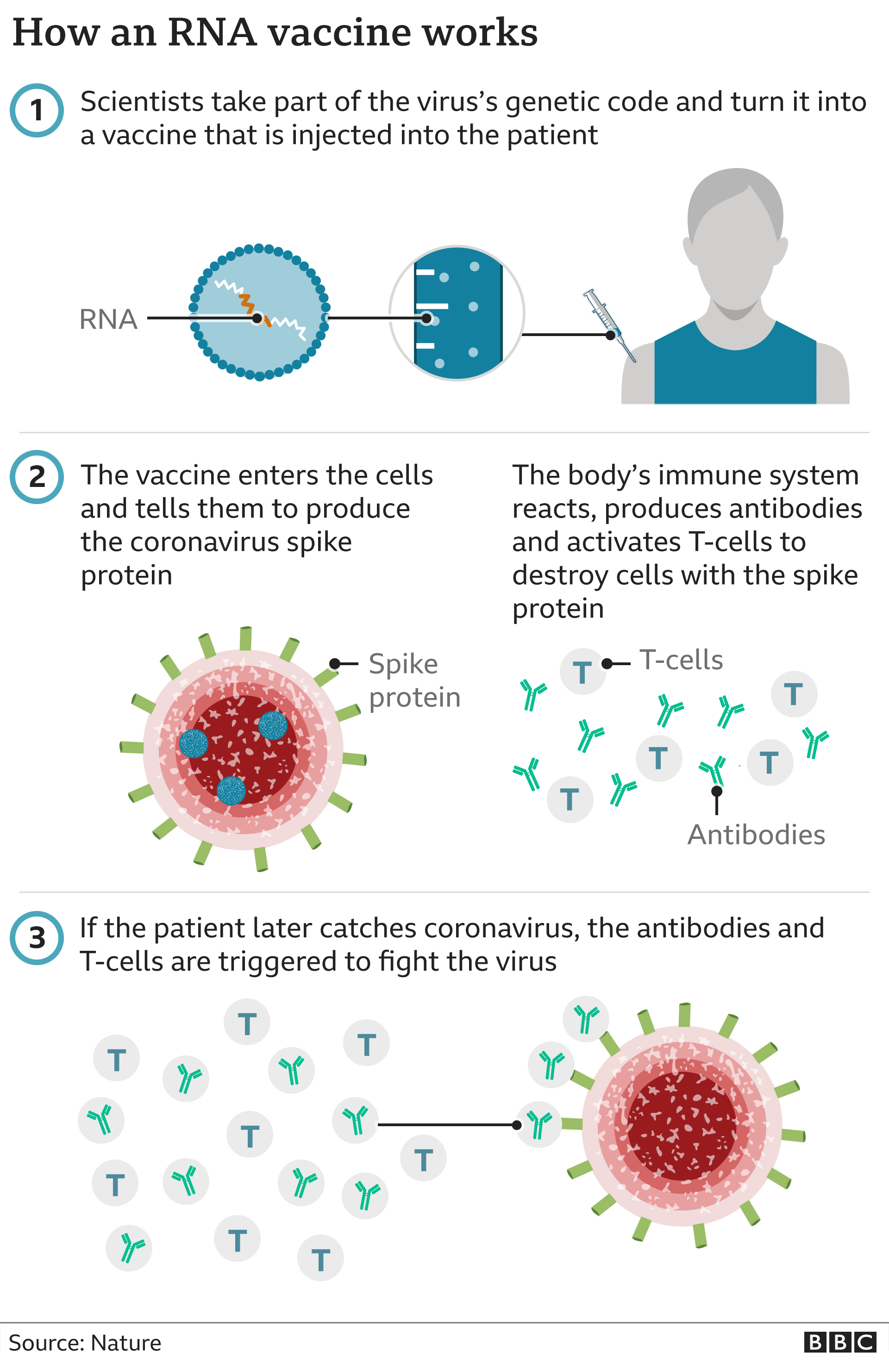
Vaccines are complex biological products with lengthy manufacturing and control processes. Vaccine manufacturing is a complex process that shepherds sensitive biological material from the lab to the vial and beyond. On average, it takes between 12-36 months* to manufacture a vaccine before it is ready for distribution. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
